News
REPLAY: METRINO Academy Session 5 “Unlocking Surface Chemistry in Nanomedicine”
What is really on the surface of your nanoparticle?
In nanomedicine, surface chemistry is where material design starts to meet biological function. It can shape stability, targeting, biocompatibility, circulation time and biodistribution.
But knowing that surface chemistry matters is not enough. The harder question is: what exactly are you measuring when you say you measured the surface?
That was the central challenge addressed during METRINO Academy Session 5, held on 23 April 2026 with Dr Ute Resch-Genger, Head of the Biophotonics Division at BAM, and moderated by Alexandre Ceccaldi and Clémence Nadal from ETPN.
The session focused on one of the most difficult layers of nanomedicine characterisation: the quantification of functional groups and coatings on nanomaterials and nanomedicines.
The key message was practical: before choosing a method, define the measurand.
A value for surface functionalisation is never just a number. It depends on the measurand, the method, the reporter, the sample preparation workflow, and the purpose of the measurement.
Missed the live session?
Watch the recording
We are pleased to share that the full recording of METRINO Academy Session 5 is now available. If you could not attend live, or if you would like to revisit the presentation and Q&A, you can now access the replay here.
With 40+ registered participants, 15 countries represented, and a satisfaction score above 8.5/10 from respondents, the session confirmed that surface chemistry is a real concern across the nanomedicine community.
The replay is especially useful for researchers, developers, analytical scientists and quality teams working on nanoparticle functionalisation, coatings, conjugation, targeting, process control, quality control, metrology or standardisation.
Live polls showed a clear need for better method selection, clearer measurands, and more robust workflows, especially as many participants reported characterising surfaces qualitatively rather than quantitatively.
Words from the expert
“If you know what you want, this could save you a lot of time.”
Dr Ute Resch-Genger, Head of the Biophotonics Division, BAM
This was a strong reminder that many challenges in surface chemistry do not come from the method itself, but from unclear expectations. Defining what needs to be measured, at which level of accuracy, and for which decision, is what ultimately determines whether a method is appropriate.

This is where surface chemistry becomes both scientifically important and analytically difficult.
“The good old methods are not a bad option.”
This second point goes one step further. Surface characterisation does not always require complex or highly specialised techniques. Robust, well understood methods such as optical assays or titrations can already provide meaningful and actionable information, especially when properly validated and aligned with the measurement objective.
Together, these two ideas open directly into one of the most important distinctions discussed during the session: what exactly is being measured when we quantify surface functional groups.
Focus on: total vs accessible functional groups
A small distinction with major consequences
One of the most important messages from Ute Resch-Genger’s presentation was that functional group quantification does not start with the instrument. It starts with the measurand. A functional group, or FG, is the chemical group on the nanoparticle surface that can drive charge, reactivity, conjugation or interaction with the biological environment.
The key question is therefore not only which method to use, but what needs to be quantified: the total amount of FGs, the reporter-accessible amount, or the composition of the near-surface region.
What is being quantified?
Different approaches can give different values because they do not always probe the same measurand. This does not automatically mean disagreement. It may mean that each method is answering a different surface chemistry question.
Total amount of FGs
Useful for material description, batch comparison, ageing studies or charge-related properties.
Measures the total amount of functional groups, either accessible to very small species or determined after sample preparation such as dissolution.
Reporter-accessible amount of FGs
Useful when the question is whether a reporter or reactive label can reach and bind to surface FGs.
Measures the fraction of FGs accessible to a signal-generating reporter. Results depend on label size, labelling efficiency, adsorption, scattering and calibration.
Near-surface composition
Useful for information on elements, chemical species, binding state and valence state near the surface.
Measures element ratios in the near-surface region. Information depth, particle curvature, beam damage and sample preparation can influence interpretation.
Validated surface chemistry data
Useful when results must support comparability, reporting, standardisation or confidence across laboratories.
Requires comparison of methods using different signal generation principles, measurands and sample preparation steps on identical samples.
This distinction explains why multi-method studies are so valuable. In the BAM and NRC comparison presented by Ute, the optical Fluram assay, potentiometric back titration, qNMR and XPS were used to understand how methods correlate, where they diverge, and what each one reveals about the same aminated silica nanoparticle systems.
The practical takeaway is simple: a surface chemistry value is the result of a measurand, a signal generation principle, a sample preparation workflow and a validation strategy.
One practical message from the session: the method must match the purpose
Ute repeatedly came back to a practical decision logic. Method selection depends on the nanoparticle system, the measurand, the required uncertainty, the sample preparation workflow, and the intended use of the result.
“Really think through what you want to achieve.”
This was especially clear during the Q&A, when the discussion turned to acceptable uncertainty. If the goal is to monitor a trend or compare relative changes, the method may not need the same level of accuracy as a method intended for standardisation or regulatory confidence.
As Ute explained, the effort required increases sharply when accuracy requirements become stricter. A method used for routine process monitoring does not need to be designed in the same way as a method being prepared for an interlaboratory comparison.
In other words, surface chemistry characterisation is not only a technical choice. It is a decision making sequence.
Q&A highlights
The Q&A was one of the strongest parts of the session because it moved naturally from analytical metrology to biological function. Participants asked about ligand density, targeting, immune response, pH responsive systems, industrial workflows, interlaboratory comparisons and the bio-nano interface.
For laboratories or companies that know surface chemistry matters but do not yet have a workflow, Ute pointed to accessible starting points.
Optical assays are often a good first option because many labs already have the necessary equipment. Titrations can also be a strong starting point, and qNMR may be accessible through university facilities. XPS can be powerful, but requires specialised equipment and expertise.
When asked which method selection criterion people tend to underestimate, Ute’s answer was immediate:
“In my opinion, the sample preparation workflow.”
She gave concrete examples: smaller particles can be harder to centrifuge or remove from dispersion, particles may not dry or dissolve properly, and optical methods can be affected by scattering or non specific adsorption.
Her simplest recommendation was to test the workflow with another person, because two scientists are unlikely to make exactly the same mistake.
For optical assays, the strongest warning was to avoid ignoring scattering and background interference. If particles are strongly coloured or strongly scattering, cleavable probes, mass balance approaches or particle removal may be more appropriate.
The broader message was simple: do not keep pushing an assay that is not robust in your own lab environment.
The discussion also connected directly to standardisation.
“ILCs are, in my opinion, the only way of doing this.”
Ute explained that interlaboratory comparisons reveal issues that a single laboratory cannot see: group bias, operator differences, instrumentation effects, protocol ambiguities and hidden assumptions.
This is why ILCs often come before standards. They show whether a method is robust across people, laboratories, backgrounds and instruments.
If you would like to explore these points in more detail, the full replay is now available.
Why this session matters for METRINO
This session captured what METRINO is ultimately about: not only developing advanced measurement techniques, but building a practical culture of comparability for nanomedicine.
Surface chemistry is a clear example. It connects material design, process control, biological function and safety, but its data only become useful when the measurand is clear, the workflow is controlled, and the result can be validated across methods and laboratories.
The Q&A showed this bridge in action. Although Ute spoke from a metrology perspective, the discussion quickly moved towards nanomedicine questions: targeting, ligand density, immune response, pH triggered release and bio-nano interactions.
Across the Academy series, each session has focused on one practical challenge. This final technical webinar showed how the same logic applies to one of the hardest layers of nanomedicine characterisation: surface chemistry.
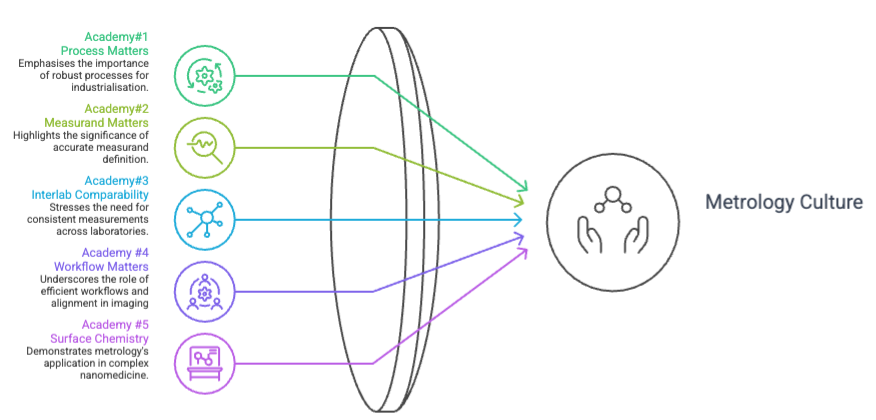
Together, the five sessions show how METRINO has translated metrology into a more practical culture for nanomedicine: from process robustness and measurand definition to interlaboratory comparability, workflow discipline and surface chemistry.
From Academy insights to community recommendations
This session closes the technical webinar sequence of the METRINO Academy, but the conversation does not stop here. The next step is to turn the lessons from the Academy and the wider METRINO project into practical recommendations for the nanomedicine community.
METRINO Final Stakeholder Session
The final stakeholder session will bring together researchers, metrology experts, industry representatives, regulators and standardisation-oriented stakeholders to discuss what should happen next for nanomedicine characterisation.
- Which METRINO outputs should be taken up by the community?
- Where are validated methods, SOPs and reference materials most needed?
- How can comparability and reproducibility become easier in daily lab practice?
- What recommendations should METRINO leave for future projects, laboratories and standardisation efforts?
About the METRINO Academy
The METRINO Academy is a short webinar series designed to translate METRINO outcomes into practical, user oriented guidance for the broader community, with a focus on real life decisions such as quality control, reproducibility and standardisation readiness.
If you missed the previous sessions, you can also revisit the replay articles for:
- Session 1 on the industrialisation of nanomedicines with Nanobiotix
- Session 2 on choosing the right measurement method
- Session 3 on RNA LNP analytics and interlaboratory comparison
- Session 4 on correlative imaging for nanomedicine
Join our growing METRINO community on LinkedIn and connect with researchers, industry and regulators working on nanomedicine metrology.